Structure of water molecule in the gas phase
A molecule of water consists of two hydrogen atoms joined to an oxygen atom by covalent bonds.
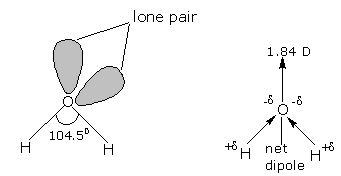
The oxygen atom has six electrons (1s2 2s2 2p4) in its outermost shell. The ‘s’ and ‘p’ orbitals of the valence shell are sp3 hybridized to form four sp3 hybrid orbitals oriented tetrahedrally around the oxygen atom. Two of the hybrid oribtals are singly occupied, while the lone pairs of electrons occupy the other two. Each singly occupied sp3 orbital overlaps with the half-filled 1s orbital of ‘H’ atom. Thus, oxygen is bonded to the two hydrogen atoms by two O-H covalent bonds, and there are two lone-pairs of electrons on the oxygen atom. Due to the presence of two lone-pairs of electrons on the O atom, the H-O-H bond angle is 104.5°, which is slightly less than the tetrahedral angle of 109°28′. Therefore, the structure of water molecule is an angular or bent structure.
Polarity of water molecule
Oxygen is more electronegative than hydrogen, being only second to fluorine in its electronegativity. Its high electronegativity causes the oxygen atom to pull the shared pairs of electrons more towards itself. As a result, the O-H bond acquires polarity. Since the two O-H bonds in water are inclined at an angle, hence the net dipole moment of water molecule is not zero. The actual dipole moment of water molecule is 1.84 debye, (denoted as D) is the unit of dipole moments.
Structure of water molecule in the liquid state
In liquid water, water molecules are held together by inter-molecular hydrogen bonds. Each oxygen atom can form two hydrogen bonds utilizing both the lone pairs on it. Experimental studies suggest that liquid water consists of aggregates of varying number of water molecules held together by hydrogen bonds and ‘free’ water molecules in a dynamic equilibrium. The aggregates continually forming, collapsing and reforming. Thus, in liquid water, the equilibrium

Structure of water in solid state (ice)
Depending on the conditions for freezing of water, the solid form of water i.e., ice, can exist in different crystalline forms. In ‘normal’ hexagonal ice, four other oxygen atoms tetrahedrally surround each oxygen atom. One hydrogen atom lies in between each pair of oxygen atoms. Thus, each and every hydrogen atom is covalently bonded to a oxygen atom and linked to another oxygen atom by a hydrogen bond. This arrangement leads to a packing with large open spaces and results in a lower density of ice than that of liquid water.
When ice melts some of the hydrogen bonds are broken and the water molecules become more closely packed. It results in an increase in the density of water above its melting points (273 K). Density of water attains a maximum value of 1 g/mL at 277 K (4°C); above 277 K, the density decreases due to the normal temperature effects.

Fig: 11.4 – Structure of normal hexagonal ice
There is one hydrogen along each oxygen – oxygen axis closer to one of the two oxygen atoms.
Solvent property of water
The polarity of water
Water has a simple molecular structure. It is composed of one oxygen atom and two hydrogen atoms. Each hydrogen atom is covalently bonded to the oxygen via a shared pair of electrons. Oxygen also has two unshared pairs of electrons. Thus there are 4 pairs of electrons surrounding the oxygen atom, two pairs involved in covalent bonds with hydrogen, and two unshared pairs on the opposite side of the oxygen atom. Oxygen is an “electronegative” or electron “loving” atom compared with hydrogen.
Water is a “polar” molecule, meaning that there is an uneven distribution of electron density. Water has a partial negative charge ( ) near the oxygen atom due the unshared pairs of electrons, and partial positive charges ( ) near the hydrogen atoms.
An electrostatic attraction between the partial positive charge near the hydrogen atoms and the partial negative charge near the oxygen results in the formation of a hydrogen bond as shown in the illustration.
The ability of ions and other molecules to dissolve in water is due to polarity. For example, in the illustration below sodium chloride is shown in its crystalline form and dissolved in water.

Many other unique properties of water are due to the hydrogen bonds. For example, ice floats because hydrogen bonds hold water molecules further apart in a solid than in a liquid, where there is one less hydrogen bond per molecule. The unique physical properties, including a high heat of vaporization, strong surface tension, high specific heat, and nearly universal solvent properties of water are also due to hydrogen bonding. The hydrophobic effect, or the exclusion of compounds containing carbon and hydrogen (nonpolar compounds) is another unique property of water caused by the hydrogen bonds. The hydrophobic effect is particularly important in the formation of cell membranes. The best description is to say that water “squeezes” nonpolar molecules together.
- at pH 7.0, a solution is neutral
- at lower pH (1-6), a solution is acidic
- at higher pH (8-14), a solution is basic
TESTING FOR WATER PURITY
- It is possible to do some simple tests to detect certain ions in water, though these tests described, suitable for doing in schools and colleges, are not effective in detecting very low concentrations.
- Never-the-less you can do simple precipitation tests e.g.
- using barium chloride to detect sulfate ions,
- and using silver nitrate to detect halide ions – chloride, bromide and iodide.
REVISION EXERCISES (POST ANSWERS USING QUESTION BOX BELOW FOR EVALUATION AND DISCUSSION. ADD QUESTION TITLE)
- What causes hardness of water?
- Mention two types of hardness of water.
- Mention two ways of removing temporary hardness of water.
- What causes permanent hardness of water?
- List two advantages of hard water.
- List two disadvantages of hard water.
- Describe two ways of testing for water in the laboratory.
- Give an equation to show what happens when slaked lime is added to temporary hard water.
Read our disclaimer.
AD: Take Free online baptism course: Preachi.com 
Free Science simulations at Classadapt (UK) Classadapt.com