CONTENT
Benzene, structure, properties and Uses
Derivative of Benzene
Aromatic Hydrocarbons
These are hydrocarbons that have the same structure as benzene.
Benzene: Benzene is a typical aromatic compound with molecular formula of C6H6.
Evaluation
- Describe three (3) ways of preparing benzene.
- Draw the structure of benzene.
Physical properties
1. It has a pleasant odour.
2. It has boiling point of 80oC.
3. Benzene can dissolve in water.
4. It burns with sooty flame.
Now playing Naming Aromatic Compounds Benzene and Phenyl in Organic Chemistry
Uses.
1. It is used as a solvent to dissolve organic.
2. It is used as fuel in petrol.
3. It is used in the manufacture of aromatic compound e.g. benzoic acid.
Evaluation
1. State two (2) uses of benzene
2. Identify two (2) chemical properties of benzene with examples.
Derivatives of Benzene
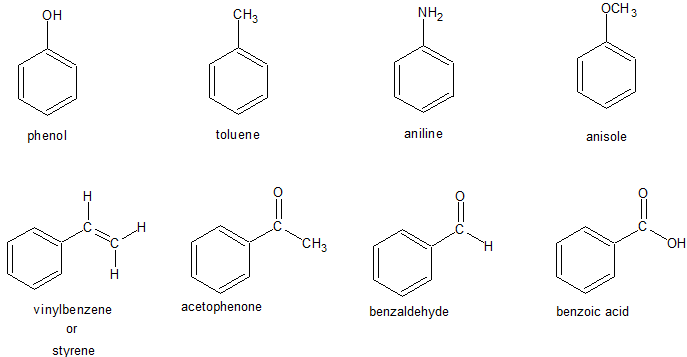
Evaluation
1. Give another name to the following compounds
(a ) Phenol (b) Toluene
2. State four (4) derivatives of benzene.
READING ASSIGNMENT
New School Chemistry By .O.Y. Ababio, pg 492-494.
WEEKEND ASSIGNMENT
1. Which of the following is the structure of benzene
2. Benzene can be prepared from the following except
(a) Coal tar (b) petroleum ( c) Alkanol (d) Ethyne
3. Benzene can undergo additional reaction due to presence of
(a) double bonds (b) single bonds ( c) hydrogen ( d) carbon.
4. Benzene undergoes the following reaction except.
(a) substitution (b) addiction ( c) Hydrogenation ( d) polymerization
5. The technique used in separating a mixture of common salt and water is
(a) evaporation (b) sublimation (c) decantation (d) chromatography.
Theory
1. a. State two (2) uses of Benzene
b. identify two (2) physical properties of benzene.
2. a. How would you prepare benzene?
b. State two (2) chemical properties of benzene.
Read our disclaimer.
AD: Take Free online baptism course: Preachi.com 
Free Science simulations at Classadapt (UK) Classadapt.com