STATES OF MATTER
Matter is made up of very tiny particles such as atoms, molecules and ions. Matter exists in three physical states namely; solid, liquid and gas. Matter has mass and occupies space.
States of Matter
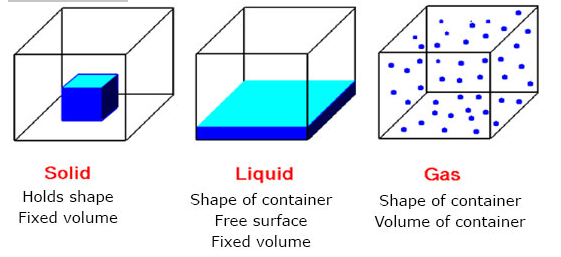
(i) Solid: The tiny particles in solid are packed very closely together hence they cannot move about. The presence of forces of attraction among the tiny particles of solid made them to be very closely packed together and allowing only vibration among them instead of moving about. This is what makes the solid to have a fixed shape and volume with high density. However, at high temperature, the molecules gain more energy and break the forces of attraction among the particles thereby causing the particles to move faster. As the temperature increases more and more, the solid melts and turns to liquid. Therefore, solid have fixed shape and volume, incompressible and very dense with the least kinetic energy when compared with those of liquid and gasses.
(ii) Liquid: The particles in the liquid are slightly further apart than those in a solid and there exist among them weaker forces of attraction (weaker force of cohesion); thus allowing them to vibrate rotate and move about. Liquid have fixed volume and a fixed shape but takes the shape of its container. When the liquid is heated, its temperature increases, the forces of cohesion / attraction becomes progressively weaker. Eventually, a stage is reached when the molecules acquire enough energy to escape as a gas. The temperature at which this occurs is called the boiling point of the liquid. Therefore, the liquid have fixed volume, no fixed shape, less dense and incompressible with kinetic energy relatively higher than those of solid state.
(iii) Gas: The particles in the gaseous state are very freely with large distances between them. The particles of gases have no fixed volume and shape, compressible and least dense with the highest kinetic energy when compared with particles of other states of matter.
EVALUATION
- Define the term matter.
- Enumerate the three main state of matter.
- Explain each of the states.
Post your answers on the forum for review
Read our disclaimer.
AD: Take Free online baptism course: Preachi.com 
Free Science simulations at Classadapt (UK) Classadapt.com