Nitrogen, which makes up about 78% of our atmosphere, is a colorless, odorless, tasteless and chemically unreactive gas at room temperature. It is named from the Greek nitron + genes for soda forming. For many years during the 1500’s and 1600’s scientists hinted that there was another gas in the atmosphere besides carbon dioxide and oxygen. It was not until the 1700’s that scientists could prove there was in fact another gas that took up mass in the atmosphere of the Earth.
Daniel Rutherford, a Scottish Physician, discovered Nitrogen in 1772. But it was Lavoisier, who proved that nitrogen is an element and that it was not a supporter of combustion and respiration. He called it ‘azote’ meaning ‘no life’. The name ‘Nitrogen’ was given to it by Jean Antoine Chaptal (1756 – 1832), in the year 1790.
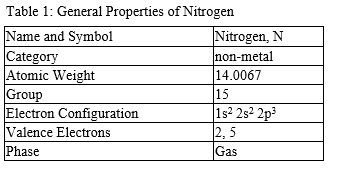
Nitrogen is found to have either 3 or 5 valence electrons and lies at the top of Group 15 on the periodic table. It can have either 3 or 5 valence electrons because it can bond in the outer 2p and 2s orbitals. Nitrogen is not reactive at standard temperature and pressure. Nitrogen is a colorless, and odorless gas that is usually found in its molecular form of (N2). For the most part, Nitrogen is inert.
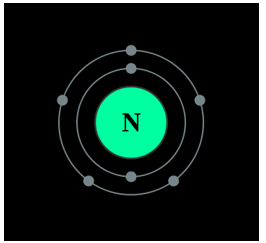
A Bohr diagram of the nitrogen atom.
Nitrogen is a non-metal element that occurs most abundantly in the atmosphere, nitrogen gas (N2) comprises 78.1% of the volume of the Earth’s air. It only appears in 0.002% of the earth’s crust by mass. Compounds of nitrogen are found in foods, explosives, poisons, and fertilizers. Nitrogen makes up DNA in the form of nitrogenous bases as well as in neurotransmitters. It is one of the largest industrial gases, and is produced commercially as a gas and a liquid.
Occurrence of Nitrogen
- Nitrogen exists in the free state and in combined form in the atmosphere of the earth.
- In the free state it occurs as N2 and occupies about 80% by volume of air.
- In the combined form, it is present as ammonia and all ammonium compounds, in nitric acid and in all nitrates, nitrites and in all living things in the form of proteins.
Isotopes
Nitrogen has two naturally occurring isotopes, nitrogen-14 and nitrogen-15, which can be separated with chemical exchanges or thermal diffusion. Nitrogen also has isotopes with 12, 13, 16, 17 masses, but they are radioactive.
- Nitrogen 14 is the most abundant form of nitrogen and makes up more than 99% of all nitrogen found on Earth. It is a stable compound and is non-radioactive. Nitrogen-14 has the most practical uses, and is found in agricultural practices, food preservation, biochemicals, and biomedical research. Nitrogen-14 is found in abundance in the atmosphere and among many living organisms. It has 5 valence electrons and is not a good electrical conductor.
- Nitrogen-15 is the other stable form of nitrogen. It is often used in medical research and preservation. The element is non-radioactive and therefore can also be sometimes used in agricultural practices. Nitrogen-15 is also used in brain research, specifically nuclear magnetic resonance spectroscopy (NMR), because unlike nitrogen-14 (nuclear spin of 1), it has a nuclear spin of 1/2 which has benefits when it comes to observing MRI research and NMR observations. Lastly, nitrogen-15 can be used as label or in some proteins in biology. Scientists mainly use this compound for research purposes and have not yet seen its full potential for uses in brain research.
Preparation of Nitrogen from Air
The chief source of free nitrogen is atmospheric air and nitrogen is usually prepared from it. Air free from dust, water vapour and carbon dioxide is compressed in a compression chamber for liquefaction.
Firstly, the pressure on the air is increased to about 200 atmospheres. It is then released through a spiral into a low-pressure area, where intense cooling of the air takes place.
Preparation of Nitrogen From Chemical Compounds
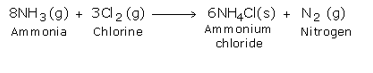
Ammonia and Ammonium Compounds
By treating excess ammonia with chlorine, ammonium chloride and nitrogen are formed.
Laboratory Preparation of Nitrogen
In the laboratory, nitrogen is prepared by heating a mixture of ammonium chloride and sodium nitrite and a small quantity of water. If ammonium nitrite is heated by itself, it decomposes to produce nitrogen gas. However, this reaction is very fast and may prove to be explosive
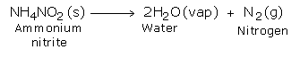
Physical Properties of Nitrogen
- Color: Nitrogen is a colorless gas.
- Odor: It has no odor.
- Taste: It is a tasteless gas.
- It provides an inter atmosphere.
- Liquid nitrogen is colorless and odorless but resembles water in appearance.
- Nitrogen exists in two allotropic forms a and b, with a transition temperature of -237° C.
- Density of nitrogen is 1.25 x 10-3 g.cm-3 at 20°C
- Melting point of nitrogen is -210 °C
- Boiling point of nitrogen is -195.8 °C
Chemical Properties of Nitrogen
Combination with elements. Nitrogen does not easily combine with other elements under ordinary conditions. A molecule of nitrogen is diatomic. These two atoms have combined by mutually sharing three pairs of electrons.
- Nitrogen forms compounds through biological activity, at high or moderate temperature in the presence of catalysts.
- It forms nitric oxide and nitrogen dioxide with oxygen.
- Its reacts with hydrogen to give ammonia.
- It gives nitrogen sulfide when treated with sulfur.
- At very high temperatures nitrogen can combine with certain active metals, such as lithium, magnesium and titanium to form metal nitrides.
- It is not a supporter of combustion.
- Energy of first ionization: 1402 kJ.mol -1
- Energy of second ionization: 2856 kJ.mol -1
- Energy of third ionization: 4577 kJ.mol -1
- Electronegativity according to Pauling: 3.0
Uses of Nitrogen
- Nitrogen is used in high temperature thermometers where mercury cannot be used.
- This is because mercury boils at 356.7oC and hence cannot be used in such thermometers.
- A volume of nitrogen is enclosed in a vessel and introduced into the region of high temperature.
- Depending upon the temperature, expansion of the nitrogen takes place.
- Then applying the gas equation, the temperature is calculated
.The Nitrogen Cyclefor the gaseous element N2(g)
- Nitrogen is an extremely important element for all plant or animal life! It is found in important molecules such as amino acids, which are combined to form proteins. Protein is used everywhere in living organisms from muscle structure in animals to enzymes in plants/animals.
- Nitrogen from the atmosphere:
- Action of nitrifying bacteria, e.g. they function in the root nodules of certain plants like peas/beans (the legumes), can directly convert atmospheric nitrogen into nitrogen compounds in plants e.g. nitrogen => ammonia => nitrates which plants can absorb.
- However, most plants can’t do this conversion from nitrogen => ammonia, though they can all absorb nitrates, so the ‘conversion’ or ‘fixing’ ability might be introduced into other plant species by genetic engineering.
- The nitrogen from air is converted into ammonia in the chemical industry, and from this artificial fertilisers are manufactured to add to nutrient deficient soils. However, some of the fertiliser is washed out of the soil and can cause pollution.
- The energy of lightning causes nitrogen and oxygen to combine and form nitrogen oxides which dissolve in rain that falls on the soil adding to its nitrogen content.
- Action of nitrifying bacteria, e.g. they function in the root nodules of certain plants like peas/beans (the legumes), can directly convert atmospheric nitrogen into nitrogen compounds in plants e.g. nitrogen => ammonia => nitrates which plants can absorb.
- N2(g) + O2(g)==> 2NO(g), then
- then 2NO(g) + O2(g)==> 2NO2(g)
- NO2(g) + water ==> nitrates(aq) in rain/soil
- Incidentally, reactions 1. and 2. can also happen in a car engine, and NO2 is acidic and adds to the polluting acidity of rain as well as providing nutrients for plants!
- Nitrogen recycling apart from the atmosphere:
- Nitrogen compounds, e.g. protein formed in plants or animals, are consumed by animals higher up the food chain and then bacterial and fungal decomposers break down animal waste and dead plants/animals to release nitrogen nutrient compounds into the soil (e.g. in manure/compost) which can then be re-taken up by plants.
- Nitrogen returned to the atmosphere:
- However, the action denitrifying bacteria will break down proteins completely and release nitrogen gas into the atmosphere.
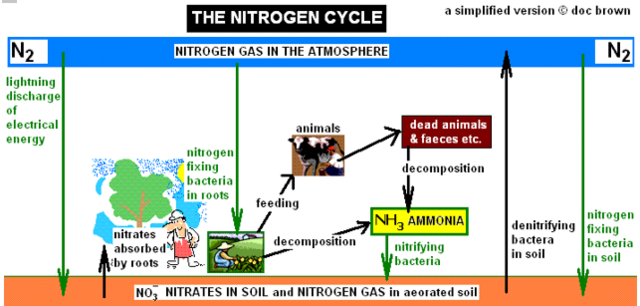
- More ‘biological detail’ of the NITROGEN CYCLE with reference to the above diagram so you can show an understanding of how nitrogen is recycled.
- a) Nitrogen gas in the air (78%, ~4/5th) cannot be used directly by most plants and all animals.
- No animals and only a few specialised plants can directly use the very unreactive nitrogen from air, but all plants nitrogen in some form to synthesise amino acids and proteins for growth and maintenance and for DNA in cell reproduction.
- However, nitrogen can be changed into nitrogen compounds like nitrates which the plants can use.
- Animals rely on plants or other animals in the food chain for their source of nitrogen compounds e.g. protein in grass, crops or other animals.
- b) Action of Nitrogen-fixing bacteria living in root nodules of plants or in the soil, their function is to fix nitrogen gas from the atmosphere into a chemical form the plant can metabolise.
- Leguminous plants like peas, lentils, clover and beans can absorb nitrogen from the air via their root nodules (swellings on the root surface) which contain enzymes capable of converting (‘fixing’) atmospheric nitrogen into soluble nitrate – a nutrient essential for amino acids, proteins and therefore plant growth.
- Legumes and their root nodule bacteria are an example of mutualism (see section 3.19 b) because the plant root supplies the bacteria with carbohydrate food and minerals and the bacteria supplies the plant in the form of the nitrate ion.
- The process of converting nitrogen in air into nitrogen compounds is sometimes called ‘nitrogen fixation‘.
- Leguminous plants like peas, lentils, clover and beans can absorb nitrogen from the air via their root nodules (swellings on the root surface) which contain enzymes capable of converting (‘fixing’) atmospheric nitrogen into soluble nitrate – a nutrient essential for amino acids, proteins and therefore plant growth.
- c) The action of lightning can convert nitrogen gas into nitrates.
- The very high electrical energy discharges from lightning activates nitrogen and oxygen molecules to react and form nitrogen oxides. These dissolve in rain to form nitrates which end up in the soil when rainwater trickles into the soil.
- d) Action of decomposers: Their function is to break down dead animals and plants
- Decomposers, e.g. various organisms like bacteria, fungi or worms can break down dead animals or plants. They break down proteins to amino acids.
- e) Action of soil bacteria: Their function is to convert proteins and urea into ammonia or ammonium ions.
- Decomposer bacteria in the soil can change proteins from dead plants/animals and urea in animal urine/droppings into ammonia/ammonium ion compounds.
- d) plus e) is sometimes called putrefaction by putrefying bacteria.
- f) Action of nitrifying bacteria: Their function is to convert this ammonia to nitrates – the process of nitrification
- Nitrifying bacteria oxidise ammonia/ammonium ions from the decayed material to form nitrates, the nitrate ion can be absorbed by plants through their root systems.
- g) Plants absorb nitrates from the soil.
- Plants absorb nitrates (soluble in water) in the moisture that the roots absorb from the surrounding soil.
- Plants can use the nitrate ion in forming amino acids from which the plant can make its proteins.
- h) Nitrates are needed by plants to make proteins for growth.
- Nitrates are an essential nutrient for plants to synthesis amino acids and hence proteins.
- i) Nitrogen compounds pass along a food chain or web of food chains.
- All food chains involve the passing of carbon compounds e.g. sugars, carbohydrates, fats and proteins up to the next trophic level i.e. the consecutive eating along a food chain (and waste produced on the way).
- e.g. grass ==> cow ==> human
- Plants make their own protein from nitrates, but animals must obtain it from plants or other animals. In fact the protein is broken down in digestion to amino acids and each animal makes its own proteins from these amino acid residues.
- All food chains involve the passing of carbon compounds e.g. sugars, carbohydrates, fats and proteins up to the next trophic level i.e. the consecutive eating along a food chain (and waste produced on the way).
- j) Action of denitrifying bacteria: Their function is to convert nitrates to nitrogen gas.
- Particular bacterial organisms can remove the oxygen from nitrate compounds to form the element nitrogen gas.
- These denitrifying bacteria live in anaerobic conditions like waterlogged soils and use the nitrate ion to respire.
- This is the opposite function of the nitrogen-fixing bacteria .
- a) Nitrogen gas in the air (78%, ~4/5th) cannot be used directly by most plants and all animals.
EVALUATION
1.Describe the physical and chemical properties of Nitrogen.
2.With the aid of a well labelled diagram,describe the preparation of Nitrogen from air
3.Describe the Laboratory preparation of a named Hydride.
4.Give an example of a reaction in which ammonia behaves as a i.reducing agent ii.base iii.precipitating agent
post your answers to the forum for review
Read our disclaimer.
AD: Take Free online baptism course: Preachi.com 
Free Science simulations at Classadapt (UK) Classadapt.com