When light falls on a metal surface, electrons are emitted, this process is called photo electric effect emission, the emitted electrons are known as photo electrons.
The maximum kinetic energy of the photo electrons are independent of the intensity of the incident light but depends on the frequency or wavelength of the incident light.
Increasing the intensity of light increases the number of photo electron but does not increase the energy or velocity. The absorbed energy is used to overcome the potential barrier of the photo electrons.
APPLICATION
Photoelectric emissions is used in the following :
I Burglary alarm
ii Television camera
iii Automatic devices for switching light at dusk e. street light.
iv. Sound production of film track
v. industrial controls and counting operations.
EINSTEN PHOTOELECTRIC EQUATION
Einstein photoelectric equation is given by
E = hf – w
W = hfo
E = maximum kinetic energy that can be given to a photo electrons
W = work function
fo = Threshold frequency
hf = W = maximum energy of the liberated. Photoelectrons.
THRESHOLD FREQUENCY (fo)
This is the lowest frequency that can cause photo emission of electrons from a metallic surface. Below threshold frequency, emission will not occur.
WORK FUNCTION (W = hfo)
This is the minimum energy required to liberate electrons from a metallic surface.
W = hfo.
EVALUATION
- If photon of wave length 2.0 X 1017m is incident on a metal and the kinetic energy of the emitted electrons is 23.5eV . Calculate the work function of the metal. (h = 6.6 x 10-34JS, 1eV = 1.6 x 10-19J, c = 3.0 x 108 ).
- Determine the threshold frequency of the metal in (1) above, hence explain what will happen if a light of frequency 9.1 x 1022Hz is illuminated on the metal.
X RAY
X-ray was discovered in 1895 by Williams Rontgen. X – rays are produced when thermally generated electrons from a hot filament are accelerated through a high potential difference and focused on to a tungsten target, where the electrons are suddenly stopped.
MODE OF OPERATION
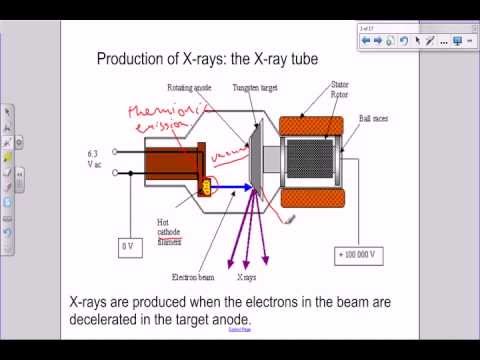
In the X- ray tube, a high potential difference is applied between the hot cathode and the anode. Electrons are emitted from the cathode and are accelerated to an extremely high speed. They are abruptly decelerated when they strike the anode causing the emission of high energy radiation of short wavelength i.e X-rays. The anode becomes very hot in the process and requires cooling gins on the outside of the tube.
ENERGY CONVERSION DURING X – RAY PRODUCTION
During X – ray production, electrical energy is converted to thermal energy. The thermal energy is converted into mechanical energy (kinetic energy) to accelerate the electron. The mechanical energy is converted into electromagnetic energy of the x-ray
TYPES OF X – RAY
There are two types of x- rays
- Hard x – rays and 2. Soft x- rays
Characteristics of Hard x-rays
- High penetrating power or ability
- Shorter wavelength
Characteristics of soft x –ray.
- low penetrating power
- longer wavelength
EVALUATION
- state the energy conversions in an x-ray tube.
- Differentiate between soft and hard x-ray.
HARDNESS
This is a measure of the strength or penetrating ability of the x – ray.
INTENSITY
This is the energy radiated per unit time per unit area by the x –ray. It depends on the current of the filament .
Properties of x- rays
- X – rays are electromagnetic waves of high frequency
- X – rays have short wavelength ( 2 x 10-10m )
- X – rays have high penetrating power
- X-rays travels in straight line
- They are not diffracted by electric or magnetic field.
- They are not diffracted by crystals.
- They ionized gases
- They cause zinc sulphide to fluoresce.
Application of X – ray
- For examining body to locate broken bones
- To detect metals and contra band in a baggage
- They are used to detect cracks n welded joints
- For investigating crystal structure
- Treatment of tumors and malignant growth
- It is used in agriculture to kill germs.
Hazards of x- rays
- It causes genetic mutation
- It can destroy body cells
- it causes leukemia, by damaging body tissues
- it causes skin burns and cancer.
Precautions
Those who work with x-rays should put on lead coat and they should always go for regular medical check-up.
GENERAL EVALUATION
- What is the function of lighting conductors
- Explain lighting
WEEKEND ASSIGNMENT
1. Which of the following give rise to the line spectra observed in atoms? (a) excitation of electrons in the atom (b) change of an electron from a higher to a lower energy level
(c) Distributed photo in the nucleus
2. Which of the following is called photo electric effect. (a) two electrons are created from a quantum of light (b) metals absorbs quanta of light and then emits electrons (c) a high energy
emits photon as it is slowed down
3. The minimum frequency that can cause photo emission of electrons from metal surface is known as (a) wavelength (b) threshold frequency (c) frequency of the incident light
4. The maximum kinetic of the photo electrons depend on (a) work function (b) frequency
(c) intensity of the incident ray
5. The minimum energy required to liberate an electron from a metallic surface is (a) ionization energy (b) work function (c) kinetic energy,
THEORY
1. (a) explain the terms “ hardness” and “ intensity” as applied to x-ray tube.
(b) State three uses of x-rays
- Determine the frequency of the photon whose energy is required to eject a surface electron with a kinetic energy of 1.970 x 10-19 eV. If the work function of the metal is 1.334 x 10-19eV.(1eV = 1.6 x 10-19J, h = 6.6 x 10-34Js, C = 3 .0 x 108ms-1)
Read our disclaimer.
AD: Take Free online baptism course: Preachi.com 
Free Science simulations at Classadapt (UK) Classadapt.com