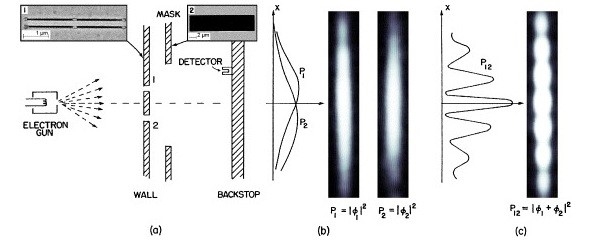
The wave nature of X-rays was established by X-ray diffraction experiments. In the same way the Davisson and Germer experiment established the wave nature of electrons.
In the Davisson and Germer experiment, a beam of electrons emitted from a heated filament was made to impinge on a layer of a thin metal film or crystal at C. The electrons were diffracted and the diffraction rings were produced on a photographic plate placed behind the thin metal film as shown below.
If the voltage V on the anode was increased, the velocity, v, of the electrons was increased. The rings were then seen to become narrower. Hence the wavelength, λ, of the electron waves decreases with increasing electron velocity.
The wavelength of a material object is given by
λ = h/mv
where mv is the momentum of the object and h is the Planck’s constant.
Later experiments showed that protons, neutrons and other particles also have the wave property of diffraction.
Read our disclaimer.
AD: Take Free online baptism course: Preachi.com 
Free Science simulations at Classadapt (UK) Classadapt.com